Leave the box blank for a nonpolar bond. Br-Br F-CH H-CI Answer Bank Bromination of isobutane is a two-step reaction.

Solved For Each Compound Determine The Direction Of Bond Chegg Com
Their bond polarity is determined according to the range it falls in.

. Solution for For each compound determine the direction of bond polarity. To determine whether a compound is poor or non polar it is important to look at the polarity of a molecule. 1 H3C----Br 2 H3C----Cu 3 Li----CH3 4 H3C----Mg----Br this could possibly have 2 arrows as there may be.
Leave the box blank for a nonpolar bond. Leave it blank for a nonpolar bond. Leave the box blank for a nonpolar bond.
Leave the box blank for a nonpolar bond. Ge-As Se-As Se-Ge The most polar bond is For each bond show the direction of polarity by selecting the correct partial charges. Looking at B S E.
For each compound determine the direction of bond polarity. As dipole formation is a characteristic of bond polarity the molecules exhibiting dipoles are more precisely called Polar covalent molecules. Experts are tested by Chegg as specialists in their subject area.
For each molecule specify the polarity of the bonds and the overall polarity of the molecule _____bonds _____molecule. Br-Br H-Cl F-CH Answer Bank. Ionic if one of the atom is a metal.
Polar covalent if both atoms are nonmetals. We review their content and use your feedback to keep the quality high. Nonpolar leave it blank.
Leave the box blank for a nonpolar bond. CHs Cн Step 1 Br- H-Br CHs CHa CH3 HyC EHo сн Step 2 Br-Br Br- CH3 CH3 HaC Overall reaction Br-Br H-Br CH A B calculate the enthalpy of each step and the enthalpy of. δSi - Pδ δSi - Sδ δS - Pδ Since silicon is the least electronegative it will have the partial positive charge in each bond.
Classify these molecules as polar or non polar H20 CCl4 BrF3. Its more likely Kohler if the bonds of the compound are not balanced out symmetrically by other poor bonds. For each bond show the direction of polarity by selecting the correct partial charges.
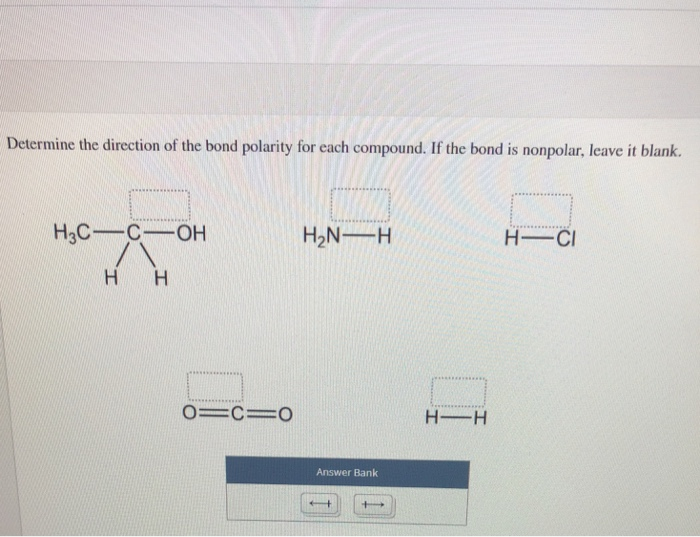
Electronegativity difference is 05 - 16. Br-Br H-CI F-CH Answer Bank. H3C-C-OH H2N-H H-CI H H.
Determine the direction of the bond polarity for each compound. Leave the box blank for a nonpolar bond. SOLVEDFor each compound determine the direction of bond polarity.
For each bond select the arrow that indicates the direction of bond polarity. For each compound determine the direction of bond polarity. KQuestion 38 of 40 For each compound determine the direction of bond polarity.
View the full answer. And since sulfur is the most electronegative it will have a partial negative charge when bonded to either silicon or phosphorus. P R is a good way to determine the true elementary in the compound to be more likely polar or non polar.
If the bond is. The polarity of the molecule is usually determined by electro negativity in its geometry. Electronegativity difference 05.
Determine the direction of bond polarity for each compound. Leave it blank for a nonpolar bond. For each compound determine the direction of bond polarity - leave a box blank for a non polar bond Br-Br H-Cl F.
In simple words a bond polarity is a scientific tool that gives us an idea about the nature of the bonds and the type of bonding they will undergo to form compounds. Leave the box blank for a nonpolar bond. Do not place an arrow for a completely nonpolar bond.
Determine the direction of bond polarity for each compound. F-CH Н-Ci Br-Br Answer Bank CGive Up. For each compound determine the direction of bond polarity.
For each compound determine the direction of bond polarity. Н-СІ F-CH3 Br-Br Answer Bank. Leave the box blank for nonpolar bondBr-BrH-CLF-CH3Answer Bank.
Br-F Br-a ___ Fa The most polar bond is For each bond show the direction of polarity by selecting the correct partial charges. Therefore the direction of polarity of each bond is as follows. For electronegativity difference 17 - 2 its.
Resources Hint 9001500 Chec Att uestion 11 of 15 If a solid line represents a covalent bond and a dotted line represents intermolecular attraction which of the choices shows a hydrogen bond. The polarity of the molecule is usually determined by electro negativity in its geometry. For each compound determine the bartleby.
For each compound determine the direction of bond polarity.
Solved Determine The Direction Of The Bond Polarity For Each Chegg Com

For Each Compound Below Click In The Box To Toggle The Direction Of Bond Polarity Leave Homeworklib

Solved For Each Compound Determine The Direction Of Bond Chegg Com

0 Comments